Isotope
From Linked Earth Wiki
Atoms can be simply described as consisting of protons, electrons, and neutrons. Isotopes of the same element differ by the number of neutrons in the nucleus, resulting in different mass.
The δ-notation
Because relative differences in isotope ratios are more precisely detected than the absolute isotopic ratios, they are commonly reported in the δ-notation:
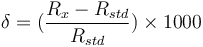
where 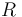 is the ratio of the abundance of the heavy to the light isotope,
is the ratio of the abundance of the heavy to the light isotope, 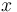 is the measured sample, and
is the measured sample, and 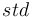 is the standard. For the element oxygen,
is the standard. For the element oxygen, 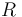 is given by 18O/16O.
is given by 18O/16O.